suitabilitycube: Simulating suitability cubes for species distribution models
The Suitability Cube (SC) is a reproducible, multidimensional structure designed to critically evaluate the outcomes of species distribution models (SDMs) in relation to the environmental data on which they are built. By organizing information across space, time, and taxa, the SC links species occurrence records with environmental predictors within a unified analytical framework, allowing a more transparent assessment of habitat suitability patterns and model reliability.
SC incorporates measures of environmental distance, which quantify how different the environmental conditions at prediction sites are from those represented in the training data. This allows identifying where model projections are environmentally supported and where they involve extrapolation into unfamiliar conditions. It also integrates metrics of niche breadth (derived from hypervolumes) to describe the ecological space occupied by each species and the Area of Applicability (AOA) to delineate the environmental domain within which model predictions can be considered valid.
By integrating these components, the SC provides a coherent system for comparing species and time periods, highlighting environmentally uncertain regions, and identifying where models can be meaningfully interpreted. Ultimately, the Suitability Cube enhances the critical evaluation of SDMs by linking what the model predicts to where those predictions should be trusted, fostering more transparent, reproducible, and ecologically grounded modelling practices.
Key concepts
Section titled “Key concepts”-
Hypervolume (HV): it represents a species’ ecological niche as an n-dimensional region defined by environmental variables, following the concept introduced by Hutchinson (1957). Each axis corresponds to an independent and ecologically relevant factor, such as temperature or precipitation, and the resulting hypervolume encompasses all combinations of conditions under which the species can persist.
-
Environmental Distance (Dissimilarity Index, DI): It quantifies how different the environmental conditions at a prediction site are from those represented in the training (occurrence) data. Following the method of Meyer & Pebesma (2021), DI is computed as a standardized distance in a multidimensional predictor space that has been (i) standardized, (ii) weighted by variable importance, and (iii) expressed relative to typical distances between training samples. This provides a unitless, comparable measure of environmental novelty
-
Area of Applicability (AOA): the Area of Applicability (AOA) defines the spatial domain where model predictions can be considered environmentally supported by the training data. It is computed directly from the Dissimilarity Index (DI) and provides a binary, spatially explicit assessment of whether a prediction site lies inside or outside the environmental space represented during model training (Meyer & Pebesma 2021).
Conceptual workflow
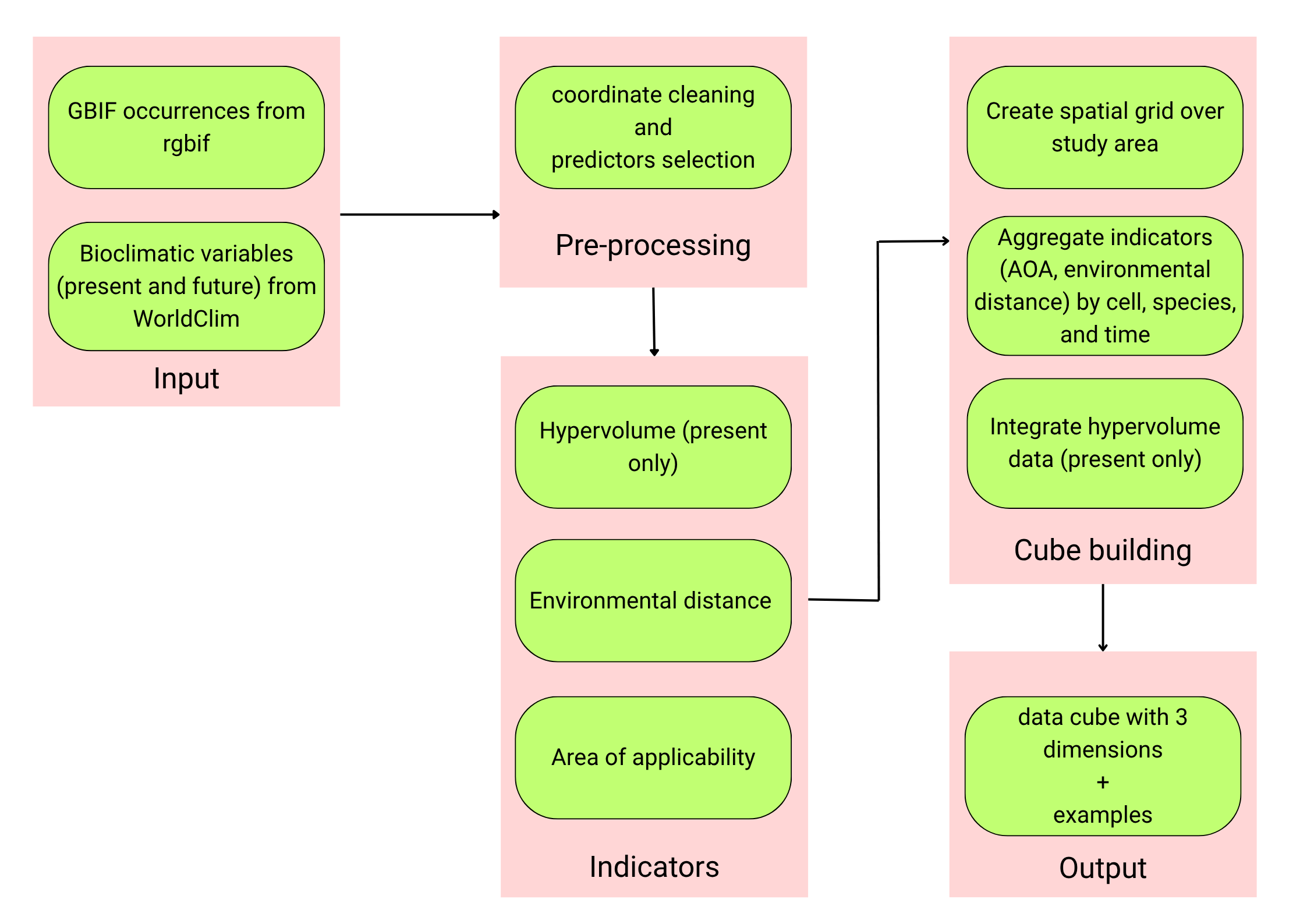
Section titled “Conceptual workflow”The conceptual workflow underlying the Suitability Cube (SC) consists of 4 main phases: data acquisition, pre-processing, indicator computation, and cube building. The workflow is organized into sequential, reproducible steps that ensure a coherent progression from data acquisition to cube construction while remaining flexible enough to accommodate different user needs and modelling contexts.
-
Data download. In the first stage, the necessary input data are collected and harmonized: the national boundary, bioclimatic predictors and species occurrence data. To reduce collinearity among predictors, a correlation-based variable selection is performed on the present dataset, and the same subset of variables is applied to future scenarios to maintain temporal consistency.
-
Pre-processing. Following data acquisition, environmental and occurrence datasets are cleaned and harmonized.
-
Indicators. This phase focuses on deriving three diagnostic indicators (HV, DI, AOA) that form the informational core of the Suitability Cube. All of them are computed from the same environmental predictors and occurrence data used to train the SDMs, ensuring consistency between model inputs and their evaluation.
-
Cube building. This phase integrates all indicators into a unified, three-dimensional structure that organizes information across space, species, and time.
Workflow
Installation
Section titled “Installation”You can install the development version of suitabilitycube from GitHub with:
Then load the package
library(suitabilitycube)Other packages needed
library(sf)library(tidyverse)library(terra)## terra 1.9.1#### Attaching package: 'terra'## The following object is masked from 'package:knitr':#### spin## The following object is masked from 'package:tidyr':#### extractlibrary(dismo)## Loading required package: raster## Loading required package: sp#### Attaching package: 'raster'## The following object is masked from 'package:dplyr':#### selectlibrary(viridis)## Loading required package: viridisLiteThis configuration defines the spatial, temporal, and taxonomic parameters used throughout the workflow.
## 1. User inputs (EDIT here)params <- list( species = c("Bufo bufo", "Bufotes viridis", "Bombina variegata"), country_name = "Italy", country_iso = "IT", res_arcmin = 2.5, ssp_code = "245", gcm_model = "BCC-CSM2-MR", period = "2041-2060", outdir = tempdir(), # change to a persistent path if desired gbif_years = c(2010, 2020), gbif_limit = 20000, cor_thr = 0.7, cor_frac = 0.10, grid_cellsize_deg = 0.25, # ~25 km grid_square = FALSE # FALSE = hex, TRUE = square)Data download
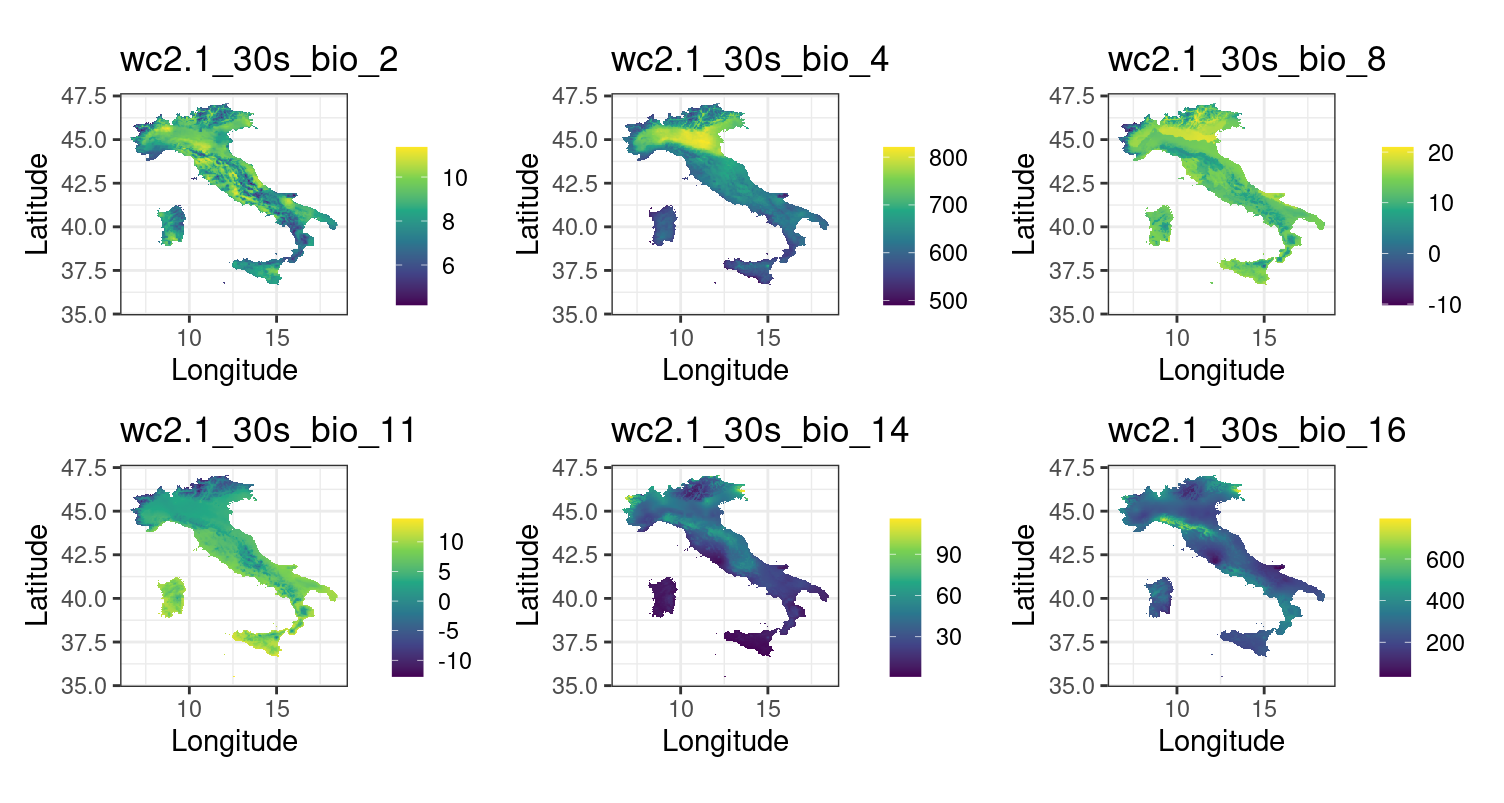
Section titled “Data download”Bioclimatic variables for the present period were downloaded from WorldClim, while those for the future were retrieved from CMIP6 for the selected Global Circulation Model (BCC-CSM2-MR), scenario (SSP245), and time window (2041–2060). Both datasets were downloaded at a spatial resolution of 2.5′ (~5 km).
After download, each dataset was cropped and masked to the national boundary of Italy, retrieved with geodata::gadm(). To guarantee spatial comparability, the present raster stack was aligned to the future using the custom helper function align_to(), which resamples the data to a common extent, resolution, and coordinate reference system.
Following alignment, a correlation-based variable selection was applied to the present bioclimatic dataset to reduce multicollinearity among predictors. Pairwise correlations were computed on a 10% random sample of grid cells, and variables exceeding a threshold of 0.7 were iteratively removed using the custom function drop_high_corr().
The resulting subset of predictors, bio3, bio4, bio8, bio11, bio14, and bio16, was retained as the final set of environmental variables. The same subset was then applied to the future dataset to ensure temporal consistency.
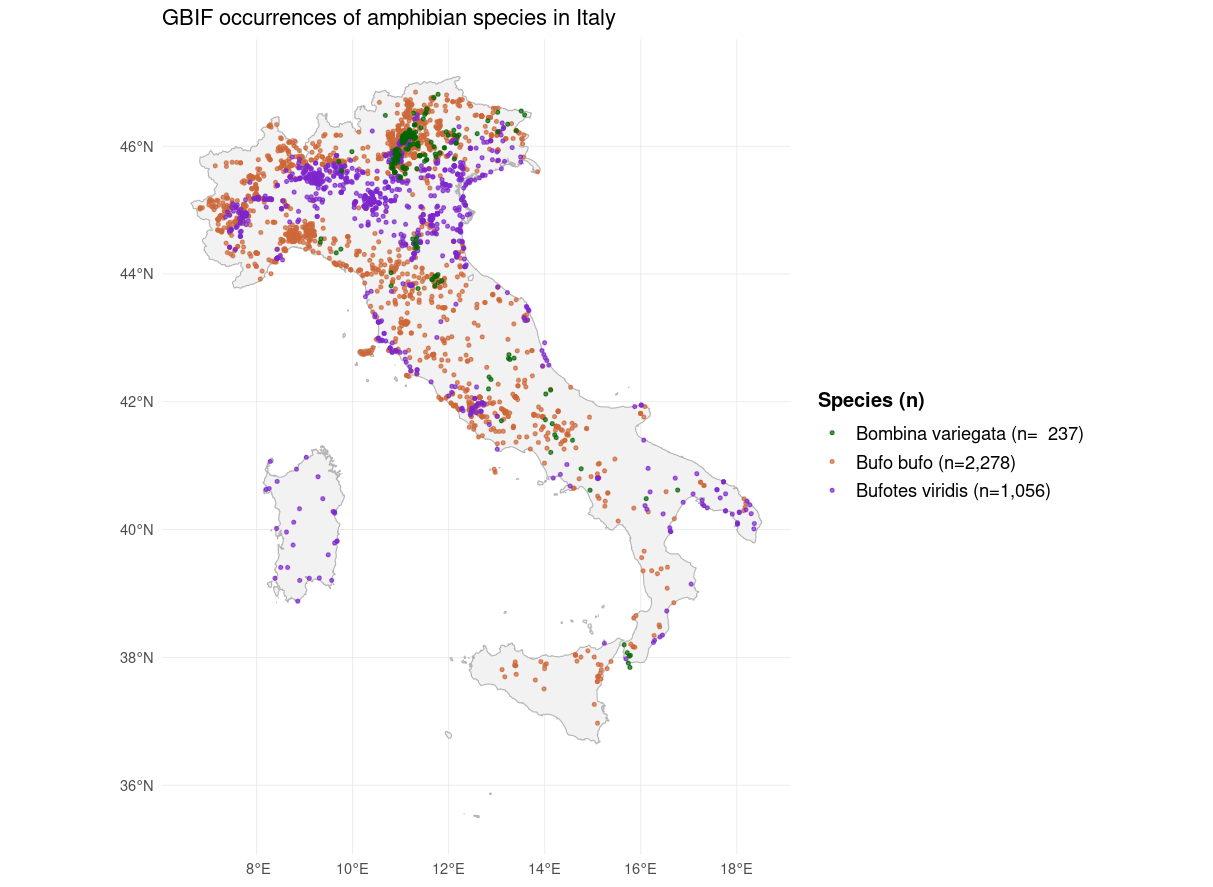
Species occurrence records were retrieved from GBIF using the R package rgbif:
- Three amphibians: Bufo bufo, Bufotes viridis, Bombina variegata)
- Country = IT,
- Years = 2010–2020,
- Record cap = 20,000 per species.
# country boundarycountry_vec <- geodata::gadm(params$country_name, level = 0, path = params$outdir)
# present bioclimatic predictorsbio_present <- geodata::worldclim_country( country = params$country_name, var = "bio", res = params$res_arcmin, path = params$outdir) |> terra::crop(country_vec) |> terra::mask(country_vec)
# future bioclimatic predictorsbio_future <- geodata::cmip6_world( model = params$gcm_model, ssp = params$ssp_code, time = params$period, var = "bio", res = params$res_arcmin, path = params$outdir) |> terra::crop(country_vec) |> terra::mask(country_vec)
# align_to aligns a raster stack to a target grid (bilinear)bio_present_aligned <- align_to(bio_present, bio_future)
## 2. Variable selection (on PRESENT)cor_res <- drop_high_corr(bio_present_aligned, thr = params$cor_thr, frac = params$cor_frac)cmat <- cor_res$corvars_keep <- cor_res$selectedvars_drop <- cor_res$dropped
# align names (do this once right after creating bio_future)names(bio_future) <- names(bio_present_aligned)
# keep same variables in the futurebio_present_sel <- bio_present_aligned[[vars_keep]]bio_future_sel <- bio_future[[vars_keep]]
## 3. GBIF occurrences --------------------------------------------------------occ_list <- gbif_occ_list(params$species, params$country_iso, params$gbif_years, params$gbif_limit)
Climatic variables
Occurrences
Hypervolume
Section titled “Hypervolume”In this workflow, the hypervolume is empirically estimated from observed species occurrences and their associated environmental predictors. The computation is performed using the R package hypervolume (Blonder et al., 2018), which applies a Gaussian kernel density estimation (KDE) method (hypervolume_gaussian) to model the probability density of species occurrences in environmental space. The hypervolume is computed only for the present period, as it depends on empirical occurrences that are not available for future conditions and could be affected by dispersal limitations or niche shifts.
-
extract_predictors_at_points: extracts raster values at point locations, keeps only selected predictor variables, removes rows with missing values, and drops numeric predictors with zero variance. -
z_transform: numeric columns are standardized (mean 0, sd 1). Missing values are replaced with the column mean prior to standardization. Columns with zero variance become all zeros -
compute_global_bw: useshypervolume::estimate_bandwidthon the predictor matrix. Returns 1 if estimation fails or produces non-finite values -
hyp_calc: computes a Gaussian hypervolume usinghypervolume::hypervolume_gaussianand returns the volume. Returns NA if there are insufficient unique points or if the computation fails -
Wrapper loop: iterates over all species to compute individual hypervolume values for the present period
AOA and DI
Section titled “AOA and DI”A set of functions was implemented to automate the computation of DI and the AOA for each species. Some functions overlap with those used for hypervolume estimation, ensuring consistency across indicators.
-
extract_predictors_at_points: extracts environmental variable values from raster layers at species occurrence locations (also used in Hypervolume computation) -
z_transform: standardizes environmental predictors to make them comparable across variables (shared with Hypervolume workflow) -
compute_aoa_pair: computes the DI for both present and future environmental rasters usingCAST::aoa(), which simultaneously derives the DI and the corresponding AOA -
Wrapper loop: iterates over all species to compute per-species DI and AOA values for the present and future periods, skipping species with insufficient training data.
Cube building
Section titled “Cube building”This phase organizes all previously computed indicators, HV, DI and AOA, into a unified, multidimensional structure that enables consistent spatial, temporal, and taxonomic analysis. The resulting cube is implemented in R using the stars package, which supports multidimensional data aligned by space, time, and species. Custom functions developed for this workflow are:
-
as_stars_on_grid: aggregates raster values to polygon grid cells and converts outputs to stars format -
build_metric_cube: builds multi-dimensional stars cubes (AOA or DI) by aggregating indicators across species and time -
build_hv_cube: generates the hypervolume cube by inserting scalar niche-size values per species -
merge_cubes: combines all indicator cubes into a single multi-attribute data cube aligned by dimensions
The process is made by the following steps, that aim to reduce the dimensionality and compress all the information in a single, reproducible object:
Output
Section titled “Output”The workflow produces a multi-attribute environmental data cube, implemented as a stars object in R.
The cube integrates the three indicators, Area of Applicability, Environmental Distance, and Hypervolume, into a single, coherent structure that supports spatial, temporal, and taxonomic analysis of model-related indicators.
The cube has three dimensions: cell, taxon, and time, corresponding respectively to spatial grid units, species, and temporal steps (present and future). Each cell contains the computed attributes.
-
AOA (binary): identifies areas within or outside the model’s environmental
-
DI (continuous): quantifies how distant local environmental conditions are from those represented in the training data
-
HV (scalar per species): describes the niche breadth, available only for the present period
# stars summary (concise)print(data_cube)## stars object with 3 dimensions and 3 attributes## attribute(s):## Min. 1st Qu. Median Mean 3rd Qu. Max. NA's## AOA 0.000 0.0000000 0.000000 0.2197779 0.0000000 1.00000 11691## DI 0.000 0.2667322 0.430247 0.4886862 0.6315494 3.36364 11691## HV 1472.595 1472.5946544 1915.109350 1776.2096349 1940.9249007 1940.92490 8232## dimension(s):## from to refsys point values## cell 1 2744 WGS 84 FALSE POLYGON ....## taxon 1 3 NA NA Bufo buf....## time 1 2 NA NA present,....Basic usage
Section titled “Basic usage”This multidimensional structure allows the exploration, visualization, and comparison of ecological indicators across locations, species, and time, while maintaining full alignment between environmental and spatial data. Some ways the data cube can be used.
Locate a cell for a given coordinate
Section titled “Locate a cell for a given coordinate”Identifies which grid cell a given geographic coordinate belongs to and displays it on a map for visual inspection.
# Define a point (lon, lat) in EPSG:4326pt <- st_sf(geometry = st_sfc(st_point(c(12.5, 42.5)), crs = 4326))
# Which grid cell contains the point?which_cell <- suppressWarnings(st_join(pt, grid_cells, join = st_intersects))if (is.na(which_cell$cell)) { message("❌ This point is NOT within the study area.") } else { cell_id <- which_cell$cell message(sprintf("Point falls inside cell #%d", cell_id)) }## Point falls inside cell #1361Basic introspection and slice
Section titled “Basic introspection and slice”Examine the cube’s organization, dimensions, and extent, enabling the inspection of selected indicators or specific subsets.
# Spatial extent of the cube (bbox of all cells)print(st_bbox(data_cube))## xmin ymin xmax ymax## 6.362442 35.280385 18.737442 47.476909# Slice example (confirm dims order): [cell, taxon, time]# AOA & DI arrays’ shapedim(data_cube[c("AOA","DI")])## cell taxon time## 2744 3 2# Cell 1361, both species, PRESENT (time = 1)data_cube[,1361, , 1]## stars object with 3 dimensions and 3 attributes## attribute(s):## Min. 1st Qu. Median Mean 3rd Qu. Max.## AOA 0.0000000 0.0000000 0.0000000 0.3333333 0.5000000 1.0000000## DI 0.1114808 0.2776988 0.4439167 0.3430515 0.4588369 0.4737571## HV 1472.5946544 1693.8520020 1915.1093496 1776.2096349 1928.0171252 1940.9249007## dimension(s):## from to refsys point values## cell 1361 1361 WGS 84 FALSE POLYGON ....## taxon 1 3 NA NA Bufo buf....## time 1 1 NA NA present# Cell 1361, both species, FUTURE (time = 2)data_cube[,1361, , 2]## stars object with 3 dimensions and 3 attributes## attribute(s):## Min. 1st Qu. Median Mean 3rd Qu. Max. NA's## AOA 0.0000000 0.0000000 0.0000000 0.000000 0.0000000 0.0000000 0## DI 0.4179193 0.4913466 0.5647739 0.562302 0.6344933 0.7042127 0## HV NA NA NA NaN NA NA 3## dimension(s):## from to refsys point values## cell 1361 1361 WGS 84 FALSE POLYGON ....## taxon 1 3 NA NA Bufo buf....## time 2 2 NA NA futureBuild a pairwise DI-difference cube (cell x comparison x time)
Section titled “Build a pairwise DI-difference cube (cell x comparison x time)”Creates a new cube that expresses the difference in environmental dissimilarity between species, per cell and time step, highlighting which species occupy more novel environmental conditions.
# Example: all pairwise differences (default)DI_diff_cube <- build_DI_diff_cube(data_cube)DI_diff_cube## stars object with 3 dimensions and 1 attribute## attribute(s):## Min. 1st Qu. Median Mean 3rd Qu. Max. NA's## DI_diff -2.35983 -0.3622762 -0.19558 -0.2231946 -0.07780134 1.792062 11691## dimension(s):## from to refsys point values## cell 1 2744 WGS 84 FALSE POLYGON ....## comparison 1 3 NA NA Bufo buf....## time 1 2 NA NA present,....Plot DI differences for a single cell
Section titled “Plot DI differences for a single cell”Visualizes pairwise DI contrasts within one location, showing which species experience greater environmental distance in the present and future.
cell_id <- 1361slice_diff <- DI_diff_cube[, cell_id, , drop = FALSE]df_diff <- as.data.frame(slice_diff) |> dplyr::select(comparison, time, DI_diff)
df_diff## comparison time DI_diff## 1 Bufo bufo - Bufotes viridis present -0.36227622## 2 Bufo bufo - Bombina variegata present -0.33243590## 3 Bufotes viridis - Bombina variegata present 0.02984032## 4 Bufo bufo - Bufotes viridis future -0.14685469## 5 Bufo bufo - Bombina variegata future -0.28629344## 6 Bufotes viridis - Bombina variegata future -0.13943875Summarize AOA coverage
Section titled “Summarize AOA coverage”Calculates the proportion of the study area that falls inside or outside the Area of Applicability for each species and time period.
aoa_df <- as.data.frame(data_cube["AOA"]) |> dplyr::select(taxon, time, AOA) |> mutate(AOA = as.integer(round(AOA))) # ensure 0/1
aoa_counts <- aoa_df |> group_by(taxon, time, AOA) |> summarise(n_cells = n(), .groups = "drop")
print(head(aoa_counts))## # A tibble: 6 × 4## taxon time AOA n_cells## <fct> <fct> <int> <int>## 1 Bufo bufo present 0 258## 2 Bufo bufo present 1 517## 3 Bufo bufo present NA 1969## 4 Bufo bufo future 0 812## 5 Bufo bufo future 1 4## 6 Bufo bufo future NA 1928Application to SDMs
Section titled “Application to SDMs”After constructing and exploring the multi-attribute environmental cube, the next step is to connect these indicators to an actual species distribution model (SDM).
We employ the R package dismo (Hijmans et al., 2023), one of the most established frameworks for building and evaluating SDMs.
We use dismo to fit a simple SDM as a proof of concept, generating a continuous suitability surface that can then be incorporated into the existing cube. The goal is not model optimization but to show how such predictions can be spatially aligned and aggregated in the same grid structure used for the AOA and DI metrics. A key advantage of this integrated approach is the ability to mask or “clip” model predictions according to the Area of Applicability (AOA). Since the AOA identifies regions of environmental space similar to those seen during model training, restricting predictions to within this area effectively filters out extrapolations beyond the model’s domain of validity.
This step transforms the SDM from a purely predictive surface into a context-aware product, in which suitability values are interpreted only where the underlying environmental relationships are supported by the data. This section illustrates:
- How an SDM output can be aligned with the cube’s spatial structure
- How it can be aggregated with the main cube
- How the AOA mask can be applied to highlight the parts of the prediction that are environmentally valid, turning the cube into a coherent framework that combines modeling, uncertainty, and applicability within a single data object
No new functions are introduced in this section. The process reuses the previously defined helper as_stars_on_grid.
SDM fitting and alignment with SC’s structure
Section titled “SDM fitting and alignment with SC’s structure”For each species (here: Bufo bufo, Bufotes viridis, Bombina variegata):
-
Extract GBIF occurrences from the previously downloaded dataset (
occ_list), and ensure they are in geographic coordinates (EPSG:4326) -
Extract coordinates (
lon,lat) from the spatial object to create a numeric matrix compatible withdismofunctions -
Fit the BIOCLIM model using the
bioclim()function, which takes a stack of environmental predictors and the occurrence coordinates as input -
Generate suitability predictions for both the present and future climate scenarios using
raster::predict(). The output is a continuous raster surface where each cell’s value ranges from 0 (unsuitable) to 1 (highly suitable).
This process is repeated for all target species, producing two raster outputs per species (present and future suitability).
library(dismo)
# climatic predictors for SDMenv_present_rs <- raster::stack(bio_present_sel)env_future_rs <- raster::stack(bio_future_sel)
# 1.1 Extract occurrence points (sf) for the speciesocc_bufo <- occ_list[["Bufo bufo"]]occ_bufotes <- occ_list[["Bufotes viridis"]]occ_bombina <- occ_list[["Bombina variegata"]]
# 1.2 Force to WGS84 lon/lat coordinates (EPSG:4326)occ_bufo <- st_transform(occ_bufo, crs = "EPSG:4326")occ_bufotes <- st_transform(occ_bufotes, crs = "EPSG:4326")occ_bombina <- st_transform(occ_bombina, crs = "EPSG:4326")
# 1.3 Extract lon/lat matrix for dismopres_xy_bufo <- st_coordinates(occ_bufo)pres_xy_bufotes <- st_coordinates(occ_bufotes)pres_xy_bombina <- st_coordinates(occ_bombina)
# 1.4 Fit a simple BIOCLIM SDM using current climate predictorsbc_bufo <- dismo::bioclim(env_present_rs, pres_xy_bufo)bc_bufotes <- dismo::bioclim(env_present_rs, pres_xy_bufotes)bc_bombina <- dismo::bioclim(env_present_rs, pres_xy_bombina)
# 1.5 Predict habitat suitability under present climatesuit_present_bufo <- raster::predict(env_present_rs, bc_bufo)suit_present_bufotes <- raster::predict(env_present_rs, bc_bufotes)suit_present_bombina <- raster::predict(env_present_rs, bc_bombina)plot(suit_present_bufo, col = viridis(100))## Error in `as.double()`:## ! cannot coerce type 'S4' to vector of type 'double'# 1.6 Predict habitat suitability under future climatesuit_future_bufo <- raster::predict(env_future_rs, bc_bufo)suit_future_bufotes <- raster::predict(env_future_rs, bc_bufotes)suit_future_bombina <- raster::predict(env_future_rs, bc_bombina)plot(suit_future_bufo, col = viridis(100))## Error in `as.double()`:## ! cannot coerce type 'S4' to vector of type 'double'Build suitability cube
Section titled “Build suitability cube”The suitability maps produced by the SDMs are then converted into a unified stars data cube. This step ensures that predicted suitability values are expressed on the same spatial grid and share the same species (taxon) and temporal (time) dimensions as the environmental indicators. The resulting cube enables consistent comparison and masking operations across indicators.
-
Define species order. The vector
species_vecis defined and checked to ensure that all species appear in the correct order across analyses -
Organize SDM outputs. Suitability rasters from the BIOCLIM models are grouped into two lists: one for present conditions and one for future conditions
-
Aggregate to the analysis grid. Each raster is aggregated over the polygon grid previously created using the helper
as_stars_on_grid -
Stack species along the taxon dimension. The aggregated suitability layers for each time period are stacked into two separate cubes, one for the present and one for the future
-
Combine time periods. The present and future cubes are merged along a new “time” dimension
-
Finalize structure. Dimension names and attribute labels are standardized, and the final cube contains a single attribute,
suitability
# 2.1 Species order (must match everything else in the project)species_vec <- params$speciesstopifnot(all(species_vec == c("Bufo bufo", "Bufotes viridis", "Bombina variegata")))
# 2.2 Put all suitability rasters into named lists (one for present, one for future)# NOTE: these are RasterLayer objects right nowsuitability_present_list <- list("Bufo bufo" = suit_present_bufo, "Bufotes viridis" = suit_present_bufotes, "Bombina variegata" = suit_present_bombina)
suitability_future_list <- list( "Bufo bufo" = suit_future_bufo, "Bufotes viridis" = suit_future_bufotes, "Bombina variegata" = suit_future_bombina)
# 2.3 Aggregate suitability to the analysis grid for each species and time.suit_present_grid_list <- lapply(species_vec, function(sp) { as_stars_on_grid( sr = terra::rast(suitability_present_list[[sp]]), grid = grid_cells, fun = mean, name = "suitability", na.rm = TRUE )})names(suit_present_grid_list) <- species_vec
suit_future_grid_list <- lapply(species_vec, function(sp) { as_stars_on_grid( sr = terra::rast(suitability_future_list[[sp]]), grid = grid_cells, fun = mean, name = "suitability", na.rm = TRUE )})
# 2.4 Stack species along a new "taxon" dimension for PRESENTsuit_present_cube <- do.call(c, suit_present_grid_list) |> stars::st_redimension() |> stars::st_set_dimensions(2, values = species_vec, names = "taxon")
# 2.5 Stack species along "taxon" for FUTUREsuit_future_cube <- do.call(c, suit_future_grid_list) |> stars::st_redimension() |> stars::st_set_dimensions(2, values = species_vec, names = "taxon")
# 2.6 Stack PRESENT and FUTURE along a new "time" dimensionsuit_cube <- c( suit_present_cube, suit_future_cube, along = list(time = c("present", "future")))
# 2.7 Make sure dimension names and attribute name are clean/standardsuit_cube <- stars::st_set_dimensions(suit_cube, 1, names = "cell")names(suit_cube) <- "suitability"
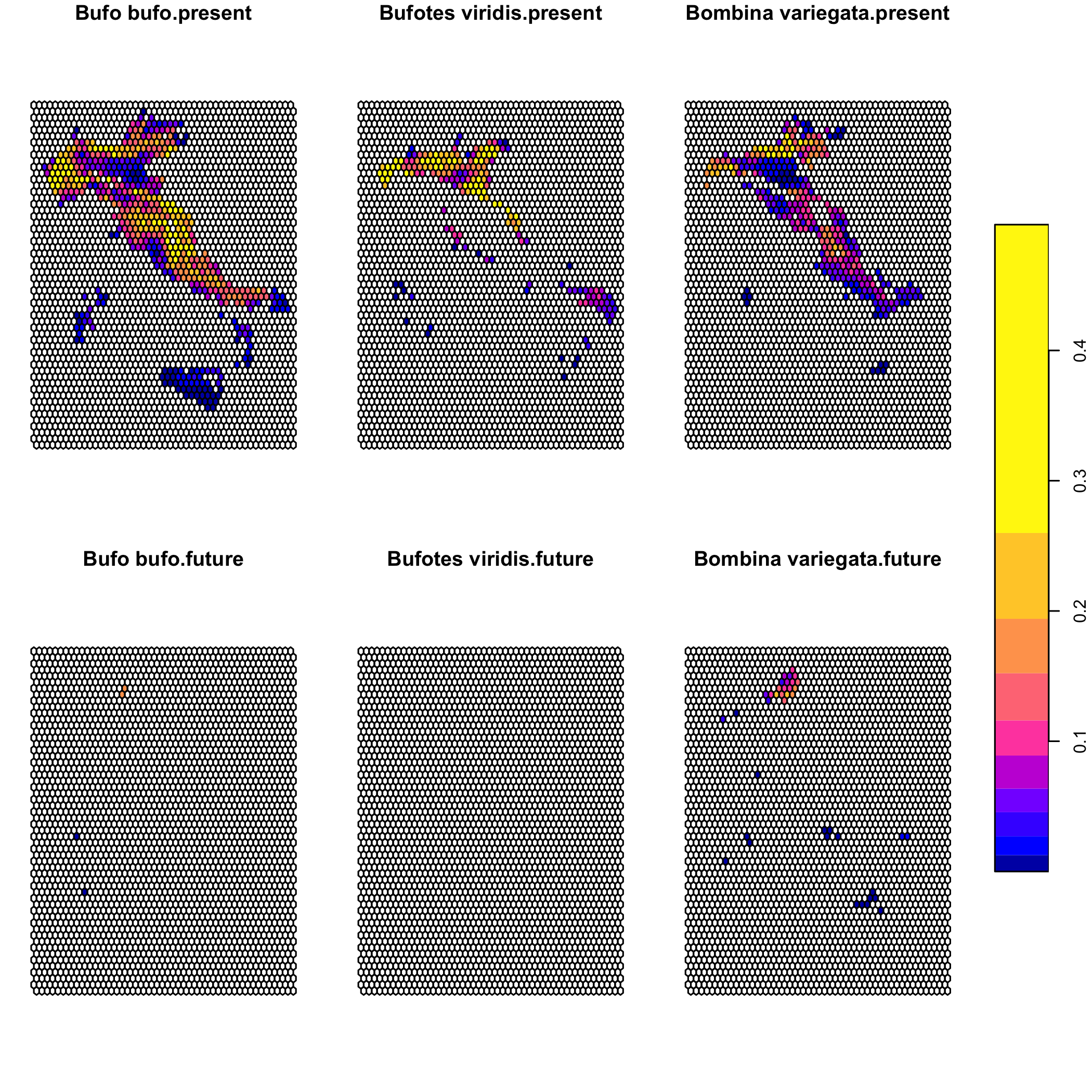
plot(suit_cube)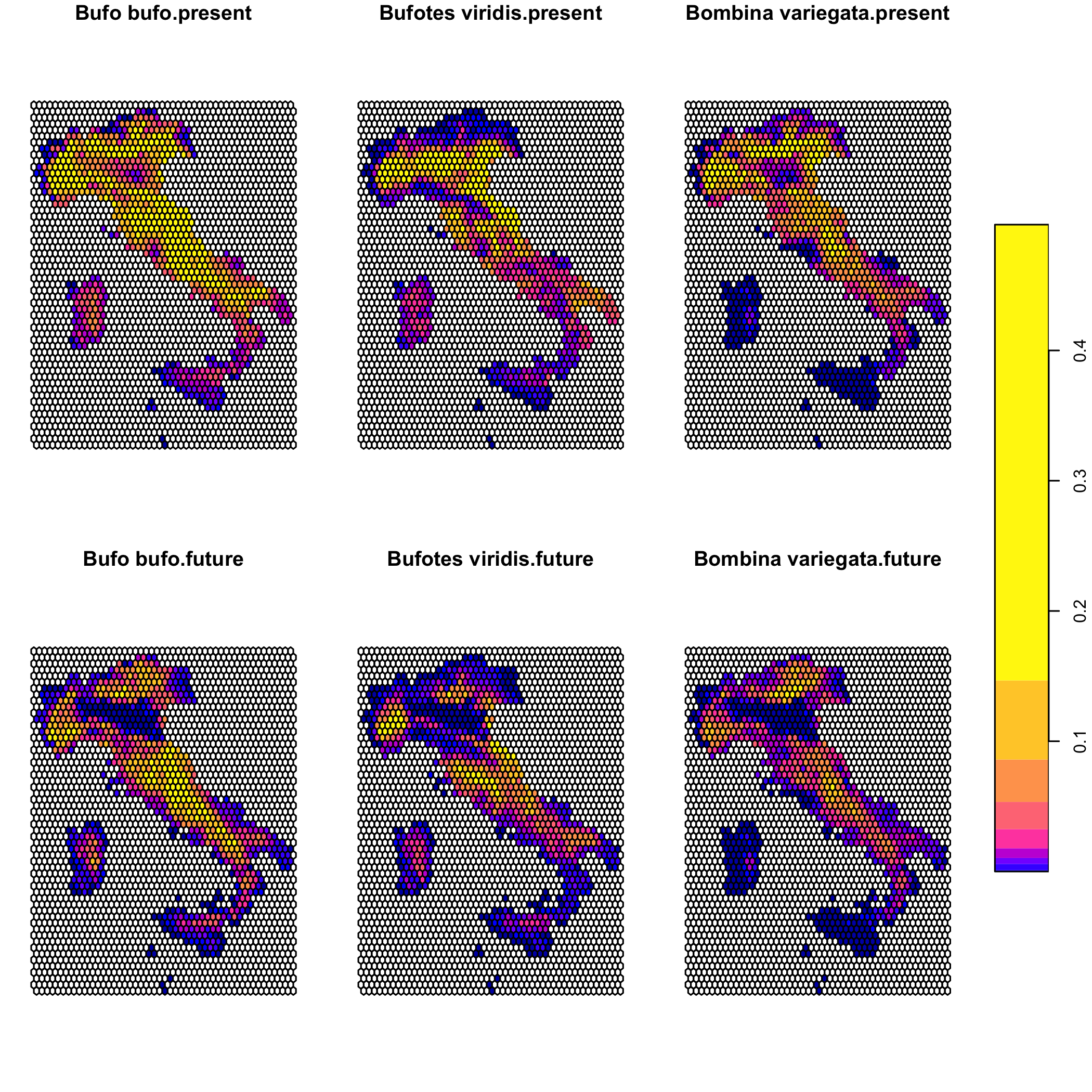
Merge suitability into the global data cube
Section titled “Merge suitability into the global data cube”After building the standalone suitability cube (suit_cube), this step integrates it into the main data_cube that already contains AOA, environmental distance (DI), and hypervolume (HV). The goal is to obtain a single multi-attribute cube where all indicators, including modeled suitability, are co-located in the same data structure.
-
Match dimensions. The taxonomic (taxon) and temporal (time) dimensions of suit_cube are explicitly aligned to those already defined in data_cube.
-
Concatenate cubes. The suitability cube is then appended to the existing data_cube using c(…).
-
Sanity check. The dimensions of the updated data_cube are inspected, and the attribute names are printed to verify that “suitability” is now part of the object.
The result is a single, enriched data_cube that stores:
-
Spatial cells
-
Species
-
Time steps
Multiple attributes: AOA, DI, HV, suitability.
This unified structure can then be queried, sliced, plotted, or summarized as one coherent object.
# 3.1 Align taxon/time labels to match the existing data_cubesuit_cube <- stars::st_set_dimensions( suit_cube, "taxon", values = stars::st_dimensions(data_cube)$taxon$values)
suit_cube <- stars::st_set_dimensions( suit_cube, "time", values = stars::st_dimensions(data_cube)$time$values)
# 3.2 Add "suitability" as a new attribute/band in data_cubedata_cube <- c(data_cube, suit_cube)
# Check that dimensions are still what we expectprint(stars::st_dimensions(data_cube))## from to refsys point values## cell 1 2744 WGS 84 FALSE POLYGON ....## taxon 1 3 NA NA Bufo buf....## time 1 2 NA NA present,....data_cube## stars object with 3 dimensions and 4 attributes## attribute(s):## Min. 1st Qu. Median Mean 3rd Qu. Max.## AOA 0.000 0.000000e+00 0.000000e+00 2.197779e-01 0.000000e+00 1.0000000## DI 0.000 2.667322e-01 4.302470e-01 4.886862e-01 6.315494e-01 3.3636395## HV 1472.595 1.472595e+03 1.915109e+03 1.776210e+03 1.940925e+03 1940.9249007## suitability 0.000 3.280481e-03 1.782178e-02 5.011102e-02 6.751238e-02 0.4965992## NA's## AOA 11691## DI 11691## HV 8232## suitability 11691## dimension(s):## from to refsys point values## cell 1 2744 WGS 84 FALSE POLYGON ....## taxon 1 3 NA NA Bufo buf....## time 1 2 NA NA present,....Apply AOA mask to SDM
Section titled “Apply AOA mask to SDM”Species distribution models often produce suitability predictions across the entire study area, including regions where extrapolation occurs beyond the environmental conditions used for model calibration. AOA identifies whether predictions fall within a known environmental space (AOA = 1) or outside it (AOA = 0).
-
Align dimensions. The taxonomic and temporal dimensions of the suitability cube (
suit_cube) are aligned with those of the AOA cube (AOA_cube) usingst_set_dimensions, ensuring element-wise correspondence between arrays. -
Apply masking. The suitability array is filtered using the AOA array: values outside the AOA (
AOA = 0or missing) are replaced withNA. -
Rebuild the masked cube. A new
starsobject,suit_cube_masked, is generated from the masked array while preserving the same spatial, taxonomic, and temporal dimensions as the original suitability cube.
The resulting suit_cube_masked retains suitability values only where the SDM operates within environmentally supported conditions. This step connects model predictions (“what the model predicts”) with their environmental validity (“where the model should be trusted”), providing a spatially explicit assessment of model transferability and reliability.
# 4.1 First, be sure suit_cube and AOA_cube share the same dimension labelssuit_cube <- stars::st_set_dimensions( suit_cube, "taxon", values = stars::st_dimensions(AOA_cube)$taxon$values)
suit_cube <- stars::st_set_dimensions( suit_cube, "time", values = stars::st_dimensions(AOA_cube)$time$values)
# 4.2 Extract raw arrayssuit_arr <- suit_cube$suitability # numeric array [cell, taxon, time]aoa_arr <- AOA_cube$AOA # 0/1 or TRUE/FALSE array [cell, taxon, time]
# 4.3 Mask: set suitability to NA wherever AoA == 0 (or AoA is NA)suit_arr_masked <- suit_arrsuit_arr_masked[ aoa_arr == 0 | is.na(aoa_arr) ] <- NA
# 4.4 Rebuild a stars object with the masked suitabilitysuit_cube_masked <- stars::st_as_stars( list(suitability_masked = suit_arr_masked), dimensions = stars::st_dimensions(suit_cube))
# outputsuit_cube_masked## stars object with 3 dimensions and 1 attribute## attribute(s):## Min. 1st Qu. Median Mean 3rd Qu. Max. NA's## suitability_masked 0 0.0365757 0.08901115 0.1156223 0.1719519 0.4965992 15415## dimension(s):## from to refsys point values## cell 1 2744 WGS 84 FALSE POLYGON ....## taxon 1 3 NA NA Bufo buf....## time 1 2 NA NA present,....plot(suit_cube_masked)